Antimicrobial Medical-Grade Vinyl for Healthcare Upholstery: Definition, Benefits, Tests & Compliance
Healthcare environments don’t choose upholstery materials the same way hotels or residential projects do. In hospitals, clinics, dental offices, and long-term care facilities, surface materials are judged by one core question:
Can they remain safe, cleanable, and stable under frequent disinfection—without becoming a hidden risk over time?
That’s where antimicrobial medical-grade vinyl becomes relevant. However, the term is often misunderstood. “Antimicrobial” is not a marketing label, and “medical-grade” is not a single universal certificate.
In real healthcare projects, what truly matters is the material system, verified laboratory testing, and complete compliance documentation proving that the surface material is suitable for long-term medical use.
This guide explains what antimicrobial medical-grade vinyl is, how it works, how it differs from ordinary synthetic leather, and which tests and compliance requirements buyers typically expect in healthcare upholstery projects.

Table of Contents
ToggleWhat Is Antimicrobial Medical-Grade Vinyl?
Antimicrobial medical-grade vinyl is a coated surface material—commonly vinyl (PVC-based) or polyurethane synthetic leather—engineered specifically for healthcare upholstery and interior applications where hygiene, durability, and chemical resistance are essential.
It combines two critical concepts:
1) “Medical-Grade” — Performance for Healthcare Use
“Medical-grade” generally indicates that the material is engineered to meet practical healthcare performance requirements, including:
-
High durability (tear strength, tensile strength, puncture resistance)
-
Resistance to repeated cleaning and disinfectant exposure
-
Stable surface appearance after frequent wiping
-
Low odor and controlled emissions (often low-VOC requirements)
-
Traceable documentation for project approvals
Importantly, medical-grade is spec-driven, meaning it is defined by the buyer’s technical requirements rather than by a single global certification.
2) “Antimicrobial” — Inhibiting Microbial Growth on the Surface
Antimicrobial functionality refers to the inclusion of antimicrobial agents within the coating system that inhibit bacterial growth on the surface under controlled conditions.
Key clarifications:
-
Antimicrobial ≠ self-sterilizing
-
Antimicrobial does not replace cleaning protocols
-
The primary value lies in reducing microbial growth between cleaning cycles, supported by laboratory testing
Why It Matters in Healthcare Upholstery
Healthcare upholstery surfaces face a combination of stresses rarely seen in other environments:
-
High-touch frequency (patients, staff, visitors)
-
Aggressive disinfection protocols (alcohol wipes, chlorine, quats)
-
Exposure to body fluids and staining risks
-
Constant abrasion from movement and equipment
-
Long service-life expectations to control maintenance cost
If surface coatings degrade—cracking, becoming sticky, fading, or peeling—the surface becomes harder to disinfect and may increase hygiene risk.
Therefore, healthcare upholstery materials must remain chemically stable, mechanically durable, and hygienic throughout their service life.
How Antimicrobial Vinyl Works (Without Over-Claiming)
Most antimicrobial vinyl relies on antimicrobial additives integrated into the coating layer. While formulations vary, the functional goal is consistent:
-
inhibit bacterial growth on the surface
-
reduce microbial colonization under defined test conditions
However, antimicrobial claims must be supported by recognized laboratory testing, not assumptions.
Commonly referenced international test methods include:
-
ISO 22196 – antibacterial activity on non-porous surfaces
-
ASTM E2180 – antimicrobial performance of polymeric materials
When antimicrobial performance is claimed, buyers typically expect:
-
clearly stated test standard
-
defined test organisms
-
full laboratory report
-
quantified antibacterial rate

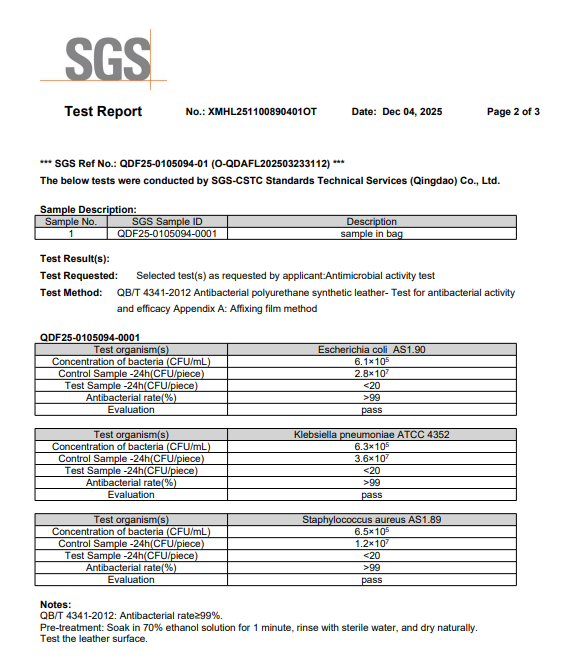
SGS-Verified Antibacterial Testing for Synthetic Leather
The antibacterial performance of this synthetic leather has been independently tested by SGS according to the following industry standard:
QB/T 4341-2012 – Antibacterial Polyurethane Synthetic Leather
This standard is specifically developed for polyurethane-based synthetic leather and coated fabrics, making it highly relevant for healthcare upholstery applications.
Test Method
-
Appendix A – Affixing Film Method
-
Direct bacterial contact on the leather surface
-
24-hour incubation under controlled conditions
-
Comparison between test sample and control sample
Tested Microorganisms
-
Escherichia coli (AS1.90)
-
Klebsiella pneumoniae (ATCC 4352)
-
Staphylococcus aureus (AS1.89)
These bacteria are commonly used indicators for hospital hygiene evaluation and are also referenced internationally.
Test Results
-
Antibacterial rate ≥ 99%
-
Test sample bacterial count: < 20 CFU
-
Evaluation: PASS
This confirms strong antibacterial performance on the synthetic leather surface under laboratory conditions.
Can Ordinary Synthetic Leather Be Used in Hospitals?
A more accurate answer than “no” is:
Not automatically—and not by default.
Many standard PU leather, PVC leather, microfiber leather, and coated fabrics are designed for furniture, automotive, or contract interiors. While visually similar, healthcare environments require additional verified performance.
To be accepted for medical upholstery, materials typically must demonstrate:
-
verified antibacterial performance (if required)
-
resistance to medical disinfectants
-
high abrasion and mechanical durability
-
chemical safety and emission compliance
-
complete technical documentation
The difference lies not in material name—but in verified performance and testing evidence.
Key Tests for Medical-Grade Upholstery Materials
1) Antimicrobial Performance
-
ISO 22196
-
ASTM E2180
-
QB/T 4341-2012 (specific to synthetic leather)
Buyers should always request full laboratory reports.
2) Chemical & Disinfectant Resistance
Critical for healthcare approval.
Materials must resist:
-
medical alcohol wipes
-
chlorine-based disinfectants
-
quaternary ammonium compounds
-
peroxide-based cleaners (facility dependent)
Acceptance focuses on:
-
no cracking or peeling
-
no surface tackiness
-
minimal color change
-
coating integrity after repeated cycles
3) Mechanical & Wear Performance
-
abrasion resistance
-
tensile and tear strength
-
puncture resistance
Ensures long service life in high-traffic medical seating.
4) Biocompatibility (When Required)
ISO 10993 may be requested for:
-
prolonged skin contact
-
specialized healthcare equipment surfaces
Requirement depends on application.
5) Fire Safety Compliance
Project-based requirements may include:
-
CA117 (United States)
-
UK BS5852 (0&1)
-
Crib 5 for high-risk seating
Fire performance depends on the complete upholstery system, not the cover material alone.

Where Medical-Grade Antimicrobial Vinyl Is Commonly Used
Healthcare Upholstery
-
hospital waiting chairs
-
clinic seating
-
dental chairs (design-dependent)
-
examination benches
Interior Surfaces
-
vinyl wallcoverings in corridors
-
coated fabrics for partitions
Long-Term Care Facilities
-
daily-clean seating
-
high-touch environments
Material Options and Selection Pathways
Healthcare upholstery materials may be engineered using different systems:
-
PVC leather / vinyl – strong disinfectant resistance and durability
-
PU leather / water-based PU / solvent-free PU / EPU leather – suitable for low-VOC, odorless, no-DMF requirements
-
Silicone leather – non-toxic positioning and chemical stability (spec-dependent)
-
Microfiber leather – premium appearance, requires validation for disinfectant resistance
Selection should always align with project specifications and test documentation.
Compliance Documentation Buyers Commonly Request
-
Technical Data Sheet (TDS)
-
SGS antibacterial test report
-
Chemical & disinfectant resistance statement
-
REACH & RoHS compliance
-
VOC / DMF / odor declarations
-
Fire test alignment (CA117 / BS5852 / Crib 5)
-
Batch traceability for large projects
In healthcare, materials are often rejected not due to performance failure—but due to incomplete documentation.
FAQ – Antimicrobial Medical Grade Synthetic Leather
1. Is the antibacterial performance laboratory tested?
Yes. The antibacterial performance has been independently tested by SGS, one of the world’s leading testing organizations.
The test follows QB/T 4341-2012 – Antibacterial Polyurethane Synthetic Leather, which is specifically designed for PU-based synthetic leather materials.
2. What antibacterial rate does the material achieve?
The tested antibacterial rate is ≥ 99%, verified after 24 hours of bacterial exposure under laboratory conditions.
3. Which bacteria were tested?
The material was tested against three common healthcare-related bacteria:
-
Escherichia coli
-
Klebsiella pneumoniae
-
Staphylococcus aureus
These bacteria are widely used indicators in medical hygiene testing worldwide.
4. Does antimicrobial leather eliminate the need for cleaning?
No. Antimicrobial performance does not replace cleaning or disinfection procedures.
Its function is to reduce bacterial growth on the surface between cleaning cycles, helping maintain hygiene stability in high-touch environments.
5. Is antimicrobial performance permanent?
The antibacterial agent is integrated into the material system rather than applied as a surface spray, allowing long-term antibacterial functionality under normal usage conditions.
6. Can the leather withstand medical disinfectants?
Yes. The material can be engineered to resist:
-
Alcohol-based wipes
-
Chlorine disinfectants
-
Common hospital cleaning chemicals
Chemical resistance can be customized based on project cleaning protocols.
7. Is this material suitable for hospital upholstery?
Yes. When combined with appropriate backing, foam, and construction methods, the material is suitable for:
-
Hospital seating
-
Clinic furniture
-
Medical waiting areas
-
Long-term care facilities
8. Is antibacterial vinyl the same as medical-grade vinyl?
Not necessarily.
Medical-grade synthetic leather requires multiple verified properties, including:
-
Antibacterial testing
-
Disinfectant resistance
-
Mechanical durability
-
Chemical safety compliance
-
Fire performance (if required)
Antibacterial testing is only one part of the full healthcare material evaluation.
9. Does antimicrobial leather comply with ISO 22196?
QB/T 4341-2012 uses similar bacterial evaluation principles to ISO 22196 and ASTM E2180 but is specifically developed for polyurethane synthetic leather surfaces, making it highly relevant for upholstery applications.
10. Can antibacterial performance be combined with fire retardancy?
Yes. The material can be customized to combine antibacterial performance with:
-
CA117 fire resistance (US)
-
BS5852 (0&1) (UK)
-
Crib 5 (contract projects)
Fire compliance depends on the full upholstery system.








